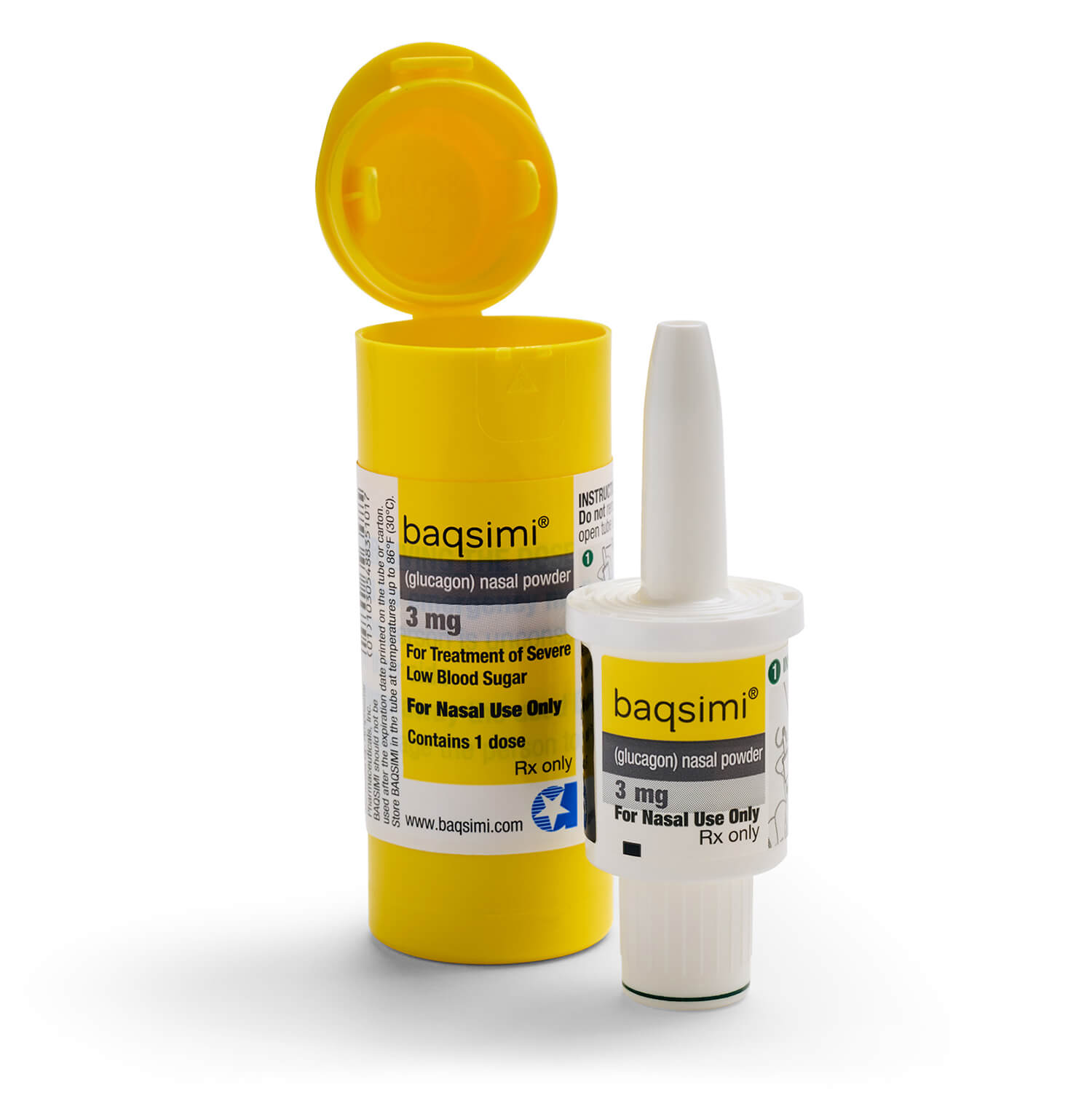
When to use Baqsimi
When to use BAQSIMI?
- BAQSIMI is used when you are having a low blood sugar emergency and are unable to eat or drink, requiring help from someone else to recover.
- It’s important that people around you recognize the possible signs of a low blood sugar, so they can be prepared to help.
- Show your family, friends, or coworkers where you keep BAQSIMI and explain how to use it by sharing these instructions. They need to know how to use BAQSIMI before you need it.
- Tell your doctor each time you use BAQSIMI. Your doctor may need to adjust your dose of drugs for diabetes.
A low blood sugar emergency can happen anytime, anywhere
For example, these episodes can happen when you miss a meal, exercise too much, do not eat enough food for the amount of insulin taken, or even at night while you sleep.
BAQSIMI is a dry nasal powder that can be given by those around you during a low blood sugar emergency.
